Electrolysis and Electroplating
Related Topics:
More Lessons for Grade 6
Math Worksheets
This is a series of lectures in videos covering Chemistry topics taught in High Schools.
In this lesson, we cover electrolysis and electroplating.
What is Electrolysis?
Definition:
A process where electrical energy drives a non-spontaneous chemical reaction, decomposing compounds into their elements or simpler substances.
Key Components:
- Electrolyte: The substance being broken down. It must contain ions (charged particles) that can move freely. This can be a molten ionic compound or an aqueous solution of an ionic compound.
- Electrodes: Conductors (usually metals like platinum, carbon, or copper) immersed in the electrolyte. They are where the electric current enters and leaves the electrolyte.
Anode: The positive electrode. Oxidation (loss of electrons) occurs here.
Cathode: The negative electrode. Reduction (gain of electrons) occurs here. - DC Power Source: A direct current (DC) power supply, like a battery or a power pack, is needed to provide the electrical energy.
How Electrolysis Works
- Ionization: Electrolyte dissociates into ions (e.g., NaCl → Na⁺ + Cl⁻).
- Movement of Ions: When a DC current is applied, positive ions (cations) in the electrolyte are attracted to the cathode, and negative ions (anions) are attracted to the anode.
Cations (Na⁺) move to the cathode.
Anions (Cl⁻) move to the anode. - Electrode Reactions: At the electrodes, the ions undergo chemical reactions:
At the Cathode (Reduction): Cations gain electrons from the cathode and are converted into atoms or molecules (gain of electrons, e.g., Na⁺ + e⁻ → Na).
At the Anode (Oxidation): Anions lose electrons to the anode and are converted into atoms or molecules (loss of electrons, e.g., 2Cl⁻ → Cl2 + 2e⁻). - Products: Elements or molecules form at electrodes (e.g., Na metal, Cl2 gas).
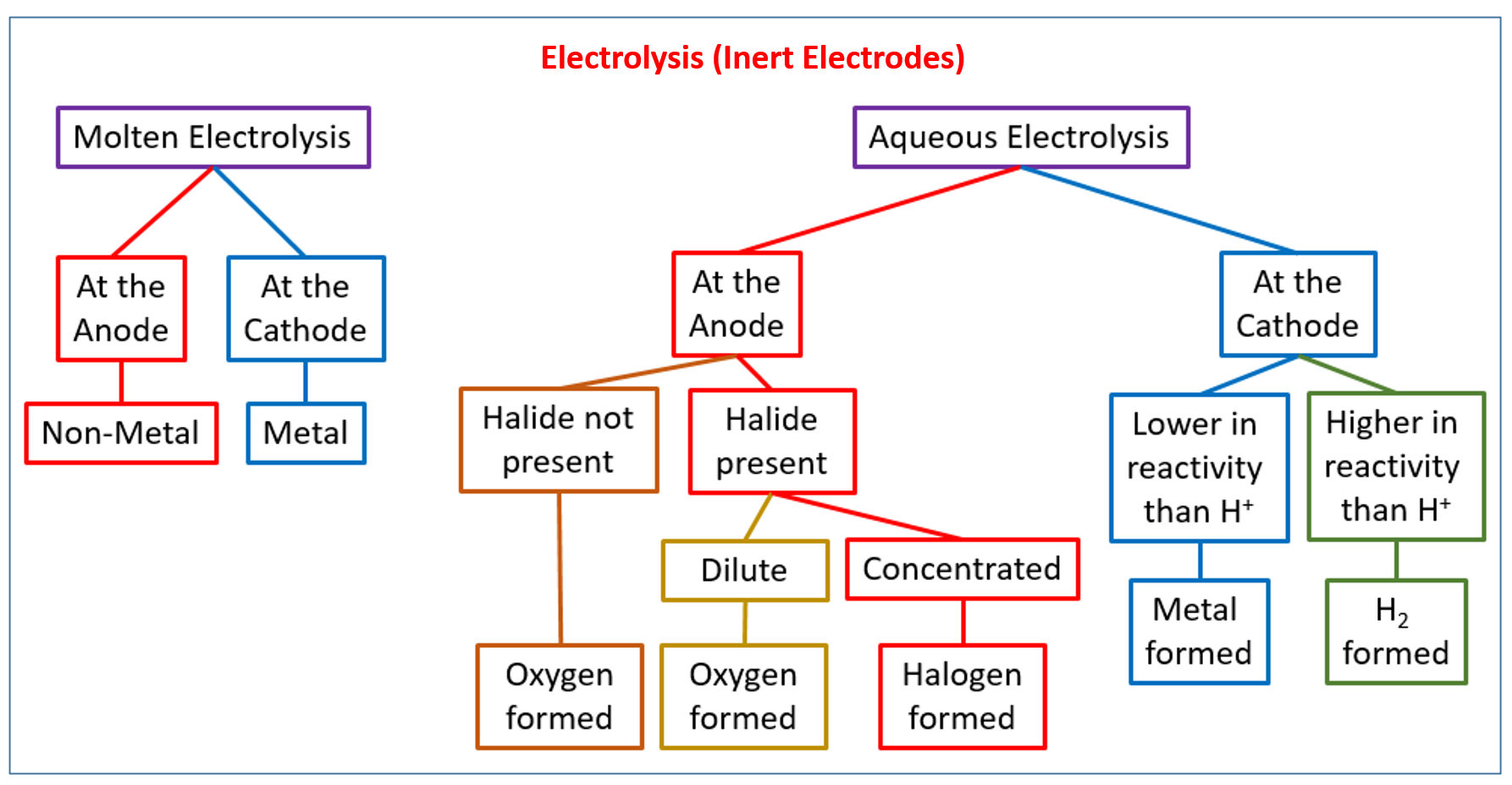
The following diagram gives a flowchart for the electrolysis of molten and aqueous compounds with inert electrodes. This flowchart is useful to determine what products will be formed at the cathode and anode for different electrolytes. Scroll down the page for examples and solutions.
Chemistry Tutorial 12.3a Electrolytic Cells:
Electrolysis Of Fused Salts
This video explains the purpose of electrolysis, and how to electrolytically decompose ionic compounds.
Chemistry Tutorial12.3b: Electrolytic Cells:
Electrolysis Of Water
This video explains the electrolysis of water.
Electroplating
Electroplating is an electrochemical process that deposits a thin layer of metal onto a conductive surface using electrolysis. It’s used for decoration, corrosion resistance, and improving conductivity.
Key Components
Anode: Made of the plating metal (e.g., copper, silver, gold). Anode dissolves (oxidation).
Cathode: The object to be plated (e.g., iron key, jewelry). Cathode gains metal (reduction).
Electrolyte: A solution containing ions of the plating metal (e.g., CuSO2 for copper plating).
Power Source: Direct current (DC) supply.
Chemistry Tutorial 12.3c: Electroplating
This video explains the purpose of electroplating one metal with another and how it works, from a chemical standpoint.
Chemistry Tutorial 12.3d: Electrolysis Demonstration
This demonstrates the electrolysis of water, sodium chloride solution and potassium iodide solution.
Try out our new and fun Fraction Concoction Game.
Add and subtract fractions to make exciting fraction concoctions following a recipe. There are four levels of difficulty: Easy, medium, hard and insane. Practice the basics of fraction addition and subtraction or challenge yourself with the insane level.

We welcome your feedback, comments and questions about this site or page. Please submit your feedback or enquiries via our Feedback page.